renal trauma


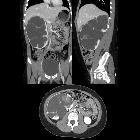











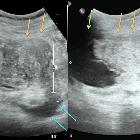





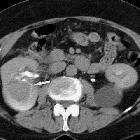


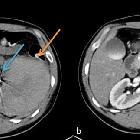

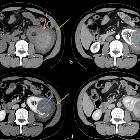
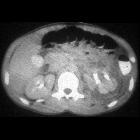
Renal trauma can result from direct, blunt, penetrating, and iatrogenic injury.
Epidemiology
Renal injuries account for ~10% of abdominal trauma, and thus the demographic of affected individuals reflect that population. The incidence of renal injuries increases in pre-existing congenital or acquired renal pathology (e.g. horseshoe kidney, renal cysts).
Clinical presentation
Patients tend to present with microscopic or macroscopic hematuria and flank and/or abdominal pain. In more severe cases, hypotension and shock may be present.
The vast majority of isolated renal trauma is minor (95-98%), the low incidence of major renal injuries is explained by the favorable anatomic position of the kidneys, which are located in the retroperitoneum.
Imaging generally should be reserved for the haemodynamically-stable patient; those who are haemodynamically-unstable are often taken directly to the operating theater.
Pathology
Etiology
Blunt trauma from motor vehicle collisions, falls, and personal collisions are the leading cause of renal injury (~85%), the mechanism is from deceleration injuries from a collision of the kidney with the vertebral column or thoracic cage.
Iatrogenic injuries can result from surgery, percutaneous renal biopsy, nephrostomy, and extracorporeal shock wave lithotripsy (ESWL).
Types
The vast majority (95-98%) of renal injuries are minor. The spectrum of renal injuries include:
- contusion/hematoma
- laceration
- hemorrhage
- avulsion of the renal pedicle leading to devascularisation of the kidney
- pseudoaneurysm
- AV fistula
- renal artery thrombosis, transection or dissection (see article: acute renal artery occlusion)
Associations
Serious renal injuries from blunt and penetrating trauma are associated with multiorgan injuries in ~80% of cases .
Radiographic features
Ultrasound
May detect hemoperitoneum but is not as accurate as CT at diagnosing renal parenchymal injuries .
CEUS
Due to the limited accessibility in the acute setting, and the lack of strong evidence (or expertise) CEUS is currently rarely used in the context of renal trauma. However, in select cases, it can be a valuable tool for problem-solving or follow-up (e.g. to reduce ionizing radiation exposure). The renal cortex demonstrates a rapid and early enhancement normally, followed by that the pyramids. Renal injuries such as lacerations appear as hypoenhancing areas, which are often wedge-shaped. Focal, patchy accumulations of contrast agent microbubbles are highly suggestive of active bleeding. If both kidneys need to be interrogated two separate contrast agent boluses are recommended. It is important to know however that CEUS does not detect collecting system injuries since the microbubbles remain intravascular .
CT
CT is the mainstay for diagnosing renal injuries:
- CT multiphase protocol study for suspected renal trauma includes a non-contrast phase, an arterial phase to evaluate the vascular injury, a nephrographic phase to evaluate renal parenchymal lesions and a delayed phase to evaluate bleeding and collecting system injuries
- an alternative protocol study is a portal venous phase followed by a delayed phase to assess for collecting system injury
See the main article renal trauma grading for a detailed description of the AAST classification of renal injuries.
Angiography
CT can provide most of the information required regarding vascular injuries, but angiography can be used to further delineate the area of injury as well as offering the opportunity for treatment with angioembolisation .
Treatment and prognosis
Treatment depends on the specific trauma and complications present.
Complications
Complications affect ~7.5% (range 3-10%) of renal injuries :
- urinoma (most common)
- delayed bleeding (within 1-2 weeks of injury)
- urinary fistula
- perinephric abscess
- hypertension from renal artery injury
- hydronephrosis
- pyelonephritis
Differential diagnosis
Renal tumors can spontaneously hemorrhage, and cause perinephric fluid collection of blood density. These include:
- renal cell carcinoma: hypervascular renal mass ± hypervascular metastases ± renal vein / inferior vena cava invasion
- angiomyolipoma: contains fat ± strong enhancement
- vasculitis (e.g. scleroderma, PAN, SLE): wedge-shaped defect / striated nephrogram; small vessels microaneurysms; capsular retraction over parenchymal lesions in chronic disease
Siehe auch:
- rupturierte Nierenzyste Trauma
- Lymphom der Niere
- Nierenblutung
- Ursachen Nierenblutung
- Nierenruptur
- renale Mikroaneurysmen bei Polyarteriits nodosa
- renale Mikroaneurysmen
- renal trauma grading
- traumatische Einblutung Nierenzyste
- Verletzungen des Nierenstiels
- Nierenblutung diabetische Nephropathie
- Makrohämaturie
- Komplikationen ESWL
und weiter:

 Assoziationen und Differentialdiagnosen zu Nierentrauma:
Assoziationen und Differentialdiagnosen zu Nierentrauma:

