heterotopic ossification




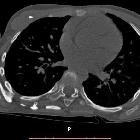

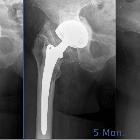
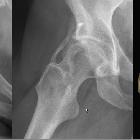




























Heterotopic ossification refers to the presence of bone in soft tissue where bone normally does not exist (extraskeletal bone). Lesions range from small clinically insignificant foci of ossification to large deposits of bone that cause pain and restriction of function.
Clinical presentation
The most common presentation is with pain around the ossification site. Associated features can include fever, soft tissue swelling, and poor mobility of the affected joint.
Pathology
The pathophysiology is unknown but most theories describe a cellular response to local tissue injury which leads to the release of chemical mediators that stimulate exaggerated bone proliferation. Muscle trauma seems to be a significant triggering event. Heterotopic ossification is histologically identical to normal cortical and cancellous bone.
Acquired heterotopic ossification can be secondary to:
- musculoskeletal trauma
- spinal cord injury
- burns
- traumatic brain injury
- postoperative
- mainly seen post joint replacement (approximately 90% of hip joint replacement patients may have heterotopic ossification )
- often forms adjacent to metal hardware
Rare genetic diseases can cause heterotopic ossification such as fibrodysplasia ossificans progressiva
Classification
The severity of heterotopic bone formation has been classified according to several systems. One traditionally used method is the Brooker classification system which divides severity into 4 types .
The Della Valle classification is a simpler modified classification system with three grades :
- grade A: absence of heterotopic ossification (may be ≥1 island of bone of <1 cm in length)
- grade B: presence of ≥1 islands of bone of at least 1 cm in length and bone spurs from the pelvis or femur; 1 cm distance between opposing bone surfaces
- grade C: bone spurs arising from the pelvis or femur with <1 cm between opposing surfaces or apparent bone ankylosis
The important distinction in reporting the presence of heterotopic ossification is therefore whether the space of between opposing bone surfaces is greater or smaller than 1 cm.
Radiographic features
Plain radiograph
The plain film is often the initial imaging investigation. Imaging features evolve as the ossification process progresses.
- early stage
- a typical finding is a soft tissue mass without calcific changes
- these can often be missed since radiographs are typically done for vague symptoms of pain
- mineralization
- can occur within 10 days after the causative insult
- calcification usually starts peripherally, though cases associated with fibrodysplasia ossificans progressiva can calcify from the central zone towards the periphery
- lesions can also be poorly organized without a recognisable mineralization pattern
- maturation
- mature cortical bone is formed if the evolving heterotopic ossification is left untreated
CT
Findings on CT mirror those of plain radiographs but CT is able to identify lesion mineralization earlier and has good overall specificity. It can sometimes be difficult to distinguish the soft tissue lesion seen early on in the evolution of heterotopic ossification from other causes and serial imaging may be required to confirm the evolution of the lesion along the typical course for heterotopic ossification.
- early stage
- low-attenuation soft tissue mass with indistinct surrounding soft tissue planes
- it may show contrast enhancement
- mineralization
- zonal mineralization pattern as described previously
- a central fatty marrow component can occasionally be seen
- maturation
- mature cortical bone at the periphery
MRI
There is no specific role for MRI once the diagnosis of heterotopic ossification has already been made. Instead, MRI is usually used in the assessment of a soft tissue mass. It has the added advantage of evaluating for other possible causes such as neoplasms (i.e. sarcoma) or underlying osteomyelitis.
- early stage
- soft tissue mass with heterogeneous high T2 signal
- lesion may manifest simply as enlargement of an involved muscle
- surrounding ill-defined high T2 signal representing edema
- enhancement of the soft tissue lesion and surrounding edematous tissue
- mineralization
- peripheral low T1 signal in the zonal pattern described
- high T1 signal centrally representing fatty marrow change
- maturation
- low T1 signal peripherally in keeping with cortical bone
- persisting T2 signal components within the lesion
- delayed
- low signal on STIR with little residual edema
See also
- myositis ossificans
- soft tissue calcification
- dystrophic calcification
- periarticular soft tissue calcification
Siehe auch:
- Myositis ossificans
- Weichteilverkalkungen
- periartikuläre Verkalkungen
- Fibrodysplasia ossificans progressiva
- periprothetische Ossifikationen
- venöse Verkalkungen
- posttraumatische Synostose
- ossifizierende Arachnoiditis
- verkalkende Myonekrose
- Heterotope Ossifikation der Membrana interossea
- neurogene heterotope Ossifikation
- Neuritis ossificans
- Brooker Klassifikation Heterotope Ossifikation
- Heterotope Ossifikation in Bauchwandnarbe
und weiter:

 Assoziationen und Differentialdiagnosen zu Heterotope Ossifikation:
Assoziationen und Differentialdiagnosen zu Heterotope Ossifikation:





