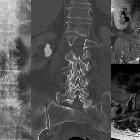
Urolithiasis



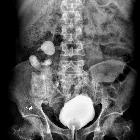





















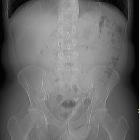























Urolithiasis refers to the presence of calculi anywhere along the course of the urinary tracts. For the purpose of the article, the terms urolithiasis, nephrolithiasis, and renal/kidney stones are used interchangeably, although some authors have slightly varying definitions of each.
See main articles: ureteric calculi and bladder stones for further discussion of these.
Epidemiology
Most patients tend to present between 30-60 years of age .
The lifetime incidence of renal stones is high, seen in as many as 5% of women and 12% of males. By far the most common stone is calcium oxalate, however, the exact distribution of stones depends on the population and associated metabolic abnormalities (e.g. struvite stones are more frequently encountered in women, like urinary tract infection as more common) .
Clinical presentation
Although some renal stones remain asymptomatic, most will result in pain. Small stones that arise in the kidney are more likely to pass into the ureter where they may result in renal colic. Hematuria, although common, may be absent in approximately 15% of patients . Strangury is also occasionally present. Some patients may also present with the complication of obstructive pyelonephritis, and may, therefore, have a septic clinical presentation.
Pathology
The composition of urinary tract stones varies widely depending upon metabolic alterations, geography, and presence of infection, and their size varies from gravel to staghorn calculi. The more common composition of stones include (more detail below):
- calcium oxalate +/- calcium phosphate: ~75%
- struvite (triple phosphate): 15%
- pure calcium phosphate: 5-7%
- uric acid: 5-8%
- cystine: 1%
- lithogenic medications: 1%
Risk factors
Certain risk factors have been identified including :
- low fluid intake
- urinary tract malformations:
- urinary tract infections
- especially with urease producing bacteria (see below)
- urease hydrolyzes urea to ammonium thus increasing urinary pH
- cystinuria: congenital disorder
- hypercalciuria: most common metabolic abnormality
- high sodium intake
- primary hyperparathyroidism
- hypervitaminosis D
- Cushing syndrome
- sarcoidosis
- milk-alkali syndrome
- hyperoxaluria
- high dietary oxalate (vegetarians)
- low gut absorption of calcium, leading to increased absorption of oxalate
- low dietary intake of calcium
- malabsorption / ileal disease (e.g. Crohn disease) resulting in fats binding calcium
- hypocitraturia
- usually idiopathic
- renal tubular acidosis (type 1)
- chronic diarrhea
- hyperuricosuria
- idiopathic/familial
- gout
- myeloproliferative disorders
- high dietary protein intake
- urinary tract diversions
Calcium-containing stones
Most renal calculi contain calcium, usually in the form of calcium oxalate (CaC2O4) and often mixed with calcium phosphate (CaPO4) . In most instances, no specific cause can be identified, although most patients have idiopathic hypercalciuria without hypercalcemia.
Brushite is a unique form of calcium phosphate stones that tends to recur quickly if patients are not treated aggressively with stone prevention measures and are resistant to treatment with shock wave lithotripsy.
Interestingly hyperuricosuria is also associated with increased calcium-containing stone formation and is thought to be related to the uric acid crystals acting as a nidus on which calcium oxalate and calcium phosphate can precipitate .
Rarely the underlying cause is primary oxaluria, a liver enzyme deficiency leading to massive cortical and medullary nephrocalcinosis, and renal failure.
Certain medications can predispose to calcium oxalate or calcium phosphate calculi, including:
- loop diuretics
- acetazolamide
- topiramate
- zonisamide
Struvite stones
Struvite (magnesium ammonium phosphate or "triple phosphate") stones are usually seen in the setting of infection with urease-producing bacteria (e.g. Proteus, Klebsiella, Pseudomonas, and Enterobacter), resulting in hydrolysis of urea into ammonium and increase in the urinary pH . They can grow very large and form a cast of the renal pelvis and calyces resulting in so-called staghorn calculi. The struvite accounts for ~70% of these calculi and is usually mixed with calcium phosphate thus rendering them radiopaque. Uric acid and cystine are also found as minor components.
Uric acid
Hyperuricosuria is not always associated with hyperuricemia and is seen in a variety of settings (see above), although in most instances uric acid stones occur in patients with no identifiable underlying etiology . Uric acid crystals form and remain insoluble at acidic urinary pH (below 5).
Cystine stones
Cystine stones are also formed in acidic urine and are seen in patients with congenital cystinuria.
Others
- medication stones :
- indinavir stones are typically radiolucent (see case 13)
- indinavir is a protease inhibitor, a class of antiretroviral drugs used in HIV treatment
- the formation of renal tract stones has since been described with other members of the protease inhibitor class
- magnesium trisilicate stones which are poorly radiopaque
- ciprofloxacin stones which are radiolucent
- sulphonamides stones which are radiolucent
- triamterene stones which are poorly radiopaque
- guaifenesin/ephedrine stones which are radiolucent
- indinavir stones are typically radiolucent (see case 13)
- pure/protein matrix stones
- mostly (~65%) made of organic proteins, carbohydrates, and glucosamines (c.f. with other stones which are crystalline with only a minor organic element)
Radiographic features
These depend on the stone composition and vary according to modality. The much greater sensitivity of CT to tissue attenuation means that some stones radiolucent on plain radiography are nonetheless radiopaque on CT.
Plain radiograph
Calcium-containing stones are radiopaque:
- calcium oxalate +/- calcium phosphate
- struvite (triple phosphate) - usually opaque but variable
- pure calcium phosphate
Lucent stones include:
- uric acid
- cystine
- medication (indinavir is best known) stones
- pure matrix stones (although may have a radiodense rim or center )
Fluoroscopy
Intravenous urography (IVU) is a traditional radiographic study of the renal parenchyma, pelvicalyceal system, ureters, and the urinary bladder. It involves the administration of intravenous contrast. This exam has been largely replaced by non-contrast CT.
Ultrasound
Ultrasound is frequently the first investigation of the urinary tract, and although by no means as sensitive as CT, it is often able to identify calculi. Small stones and those close to the corticomedullary junction can be difficult to reliably identify. Ultrasound compared to CT KUB reference showed a sensitivity of only 24% in identifying calculi. Nearly 75% of calculi not visualized were <3 mm . Features include :
- echogenic foci
- acoustic shadowing
- twinkle artifact on color Doppler
- color comet-tail artifact
Pulsed wave (PWD) and color flow Doppler (CFD) are further sonographic modalities that may act as a diagnostic aid, and assess for the presence of complications;
- ureteric jets in obstructive uropathy tend to be shorter, slower, and occur less often
- suggested cutoff values vary; the combination of fewer than 1.5 jets per minute, with peak velocities below 19.5 cm/s and jet durations less than 2.5 seconds have specificities ranging between 87 and 97%
- the renal resistive index (RI) is significantly higher in obstructed kidneys
- contralateral unaffected renal RI comparison useful
- elevation in RI may precede pelvicalyceal dilation
CT
On CT almost all stones are opaque but vary considerably in density.
- calcium oxalate +/- calcium phosphate: 400-600 HU
- struvite (triple phosphate): usually opaque but variable
- pure calcium phosphate: 400-600 HU
- uric acid: 100-200 HU
- cystine: opaque
Two radiolucent stones are worth mentioning :
- medication (protease inhibitor (indinavir)) stones
- radiolucent and usually undetectable on non-contrast CT
- characterized on delayed phase as a filling defect in the ureter
- pure matrix stones
99% of renal tract calculi are visible on a non-contrast CT. Given that one of the commonest sites for a stone to become lodged is the vesicoureteric junction, some centers perform the study in the prone position to establish if the stone is retained within the intravesical component of the ureter or has already passed into the bladder itself.
Dual-energy CT
Dual-energy CT is a technique allowing the composition of the calculus to be determined, by assessing stone attenuation at two different kVp levels. Each CT vendor has its own algorithms for the use of dual-energy CT for assessing stone composition. Dual-energy CT may be useful in detecting stones concealed by the opacification of the collecting system .
Dual-energy CT has also been shown to predict the success of extracorporeal shock wave lithotripsy .
Treatment and prognosis
Indications for surgical management include:
- larger stones, typically that above 5 mm in size
- extended duration of symptoms
- location of the stone, with proximal calculi less likely to spontaneously pass
- infection or septic features
- certain professions (airline pilot, truck driver) due to the risk of renal colic during work
- solitary kidney
- failed conservative management
Surgical intervention typically involves a retrograde ureteric stent with subsequent laser lithotripsy. In acutely septic patients who would be unsuitable for an anesthetic or in those who would not be suitable for a retrograde stent, such as those with poor retrograde access or abnormal anatomy, a percutaneous nephrostomy (PCN) with an antegrade stent followed by laser lithotripsy is preferred.
Extracorporeal shock wave lithotripsy (ESWL) is usually performed in large proximal calculi in patients unsuitable for invasive management.
Percutaneous nephrolithotomy (PCNL) is usually reserved for large calculi near the pelviureteric junction, especially staghorn calculi, which are unlikely to be removable via retrograde access.
Calcium stones
Small asymptomatic stones in the kidney can be safely ignored, and if patients maintain good states of hydration, the risk of recurrent symptoms can be dramatically reduced . In all settings, a search for a possible underlying cause of hyperoxaluria/hypercalciuria should be sought and if present corrected when possible.
Larger stones may be treated with:
Struvite stones
Struvite stones are usually large (staghorn calculi) and result from infection. These stones need to be treated surgically and the entire stone removed, including small fragments, as otherwise, these residual fragments act as a reservoir for infection and recurrent stone formation.
Uric acid stones
Uric acid stones usually are the result of low urinary pH, and hydration and elevation of urinary pH to approximately 6 are usually sufficient (note rendering the urine too alkali (e.g. pH >6.5) may result in calcium stone formation) .
Cystine stones
Cystine stones may be difficult to treat and are difficult to shatter with ESWL. Hydration and alkalinisation are usually first-line therapy.
Complications
Recognized complications include:
- spontaneous extravasation: spontaneous rupture of renal pelvis (SRRP) +/- urinoma formation (see case 23)
- recurrent urinary tract infections (pyelonephritis)
- impaction of the stone along the ureter may result in hydroureter and/or hydronephrosis
- chronic obstruction may lead to progressive renal parenchymal damage, impaired renal function and renal failure
- although rare, urolithiasis is a risk factor for squamous metaplasia and squamous carcinoma (possibly as a result of chronic local inflammation and infection)
Differential diagnosis
The differential of renal calculi is essentially that of abdominal calcifications. On CT there is usually little confusion as not only is CT exquisitely sensitive in detecting stones, but their location can also be precisely noted. If non-contrast CT is equivocal for the location of the calcification, then a repeat CT with urographic phase contrast is usually able to clarify
Thus the differential diagnosis is predominantly on plain radiograph, and to a lesser degree ultrasound:
- cholelithiasis overlying right kidney
- pancreatic calcification
- phleboliths
- calcified mesenteric lymph nodes
- renal artery calcification
- intrarenal gas (only a differential for ultrasound)
- acoustic shadow is usually 'dirtier'
- gas typically more mobile than stones
- pure/protein matrix stones may mimic an upper tract soft tissue mass

 Assoziationen und Differentialdiagnosen zu Urolithiasis:
Assoziationen und Differentialdiagnosen zu Urolithiasis: