giant cell tumor
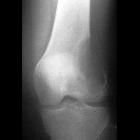
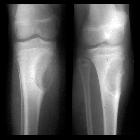
Teenager with
knee pain. AP radiograph of the knee shows growth plate fusion and a metaphyseal lesion that is lytic and expansile in appearance with a narrow zone of transition and no associated periosteal reaction.The diagnosis was giant cell tumor.

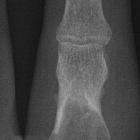
Giant cell
bone tumor in the head of the 4th metacarpal of the left hand. The location suggests an Enchondroma as differential diagnosis.

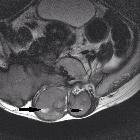
MRI imaging
of soft tissue tumours of the foot and ankle. Giant cell tumour of the tendon sheath. a Sagittal T2FS image shows a heterogeneously hyperintense lesion underlying the metatarsal, related to the flexor tendon. b Gradient echo imaging shows foci of blooming (arrowhead)

Localized
tenosynovial giant cell tumor • Tenosynovial giant cell tumor (finger) - Ganzer Fall bei Radiopaedia

Localized
tenosynovial giant cell tumor • Giant cell tumor of the tendon sheath (finger) - Ganzer Fall bei Radiopaedia
Localized
tenosynovial giant cell tumor • Giant cell tumor of tendon sheath - Ganzer Fall bei Radiopaedia
Häufige Lokalisationen von Riesenzelltumoren sind solche des Knochens und der Sehnenscheiden. Der Riesenzelltumor des Knochens wird auch als Osteoklastom bezeichnet. Der Riesenzelltumor der Weichteile (oft Sehnenscheiden) ist mit der pigmentierten villonodulären Synovialitis verwandt.
Siehe auch:
- nicht ossifizierendes Fibrom
- Aneurysmatische Knochenzyste
- Enchondrom
- Chondrosarkom
- Lucent/lytic bone lesion - differential diagnosis (mnemonic)
- intraossäres Ganglion
- Multiples Myelom
- primärer Hyperparathyreoidismus
- Chondroblastom
- Renale Osteodystrophie
- Chondromyxoidfibrom
- desmoplastisches Fibrom
- Riesenzelltumor des Knochens
- Riesenzelltumor der Sehnenscheiden
- Ostitis fibrosa cystica
- Riesenzelltumor des Sakrums
- Riesenzelltumor der Wirbelsäule
- Metastasen
und weiter:
- Tumoren der Schädelkalotte
- Codman-Dreieck
- Fibröse Dysplasie
- Cholesteatom
- Chordom
- einfache (juvenile) Knochenzyste
- solitäre lytische Läsion des Schädels
- Pigmentierte villonoduläre Synovialitis
- solide Periostreaktion
- erworbenes Cholesteatom
- Cholesteatom des äußeren Gehörgangs
- spinal myxopapillary ependymoma
- single layer periosteal reaction
- epiphysäre Knochentumoren
- spinales Ependymom
- Tumoren der Thoraxwand
- oberflächliche Weichteilläsionen der Extremitäten
- osseous lesions preferentially involving the epiphysis
- skeletal mass with fluid-fluid levels
- spinales Ependymom des Filum terminale
- differential diagnosis for metatarsal region pain
- Weichteiltumoren der Extremitäten
- lytic bone lesion (mnemonic)
- Riesenzelltumor Fingerknochen
- Plasmozytom des Knochens solitär
- Osteoklastom
- Knochenläsionen der Epiphyse
- sacrococcygeales Chordom
- primary tumours of the spine
- posterior vertebral body lesions (mnemonic)
- Riesenzelltumor des Beckenknochens
- calcaneal lesions (mnemonic)
- Riesenzelltumor der Patella
- Riesenzelltumor distales Femur
- Flüssigkeit-Flüssigkeitsspiegel
- Knochenläsionen der Metaphyse
- giant cell tumour in children
- giant cell tumor of the tendon sheath of the hand
- distal radius giant cell tumour
- Riesenzelltumor des Pankreas

 Assoziationen und Differentialdiagnosen zu Riesenzelltumor:
Assoziationen und Differentialdiagnosen zu Riesenzelltumor:Lucent/lytic
bone lesion - differential diagnosis (mnemonic)