medulloblastoma

































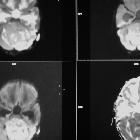


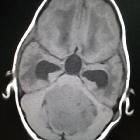
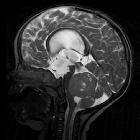



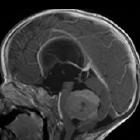











Medulloblastomas are the most common malignant brain tumor of childhood. They most commonly present as midline masses in the roof of the 4ventricle with associated mass effect and hydrocephalus. Treatment typically consists of surgical resection, radiation therapy, and chemotherapy, with the prognosis strongly influenced by surgical resection, the presence of CSF metastases at the time of diagnosis, and expression of the c-erbB-2 (HER2/neu) oncogene.
Although medulloblastoma has been classically thought of as a single entity it is becoming increasingly evident that there are a number of distinct molecular subgroups with, albeit overlapping, clinical, histological and imaging features . This has been reflected in the 2016 update of the WHO classification of CNS tumors, which recognizes four molecular subgroups, although it is worth noting that recent publications have identified up to 12 subgroups .
The 4 molecular subgroups currently recognized in the WHO classification have been named WNT, SHH, Group 3, and Group 4 . Perhaps counterintuitively what most of us think of as the typical medulloblastoma (a midline tumor in early childhood) is group 4. Further information on the subtypes of medulloblastoma is available in the links below and in the article classification of medulloblastomas.
Epidemiology
Overall medulloblastomas account for 12-25 % of all pediatric CNS tumors, and 30-40% of pediatric posterior fossa tumors . They are also seen in adults but only account for 0.4-1.0% of adult brain tumors . Since there are many more adults than children, 14-30% of all medulloblastomas are found in adults.
Taken as a group, there is a moderate male predilection with a M:F ratio of 2:1, although this is only true of group 3 and 4 tumors .
They usually present in childhood with 77% of cases before the age of 19. The median age of diagnosis is 9 years. When diagnosed in adulthood, they typically present in the 3 and 4 decades and are more likely to arise in atypical locations (see below). When they present in adulthood, there is often a better prognosis.
Importantly the age of presentation and gender ratio is influenced by tumor genomics :
- WNT (least common)
- children and adults (not seen in infancy)
- M:F 1:1
- SHH
- infants and adults (rare in children)
- M:F 1:1
- group 3
- infants and children (rare in adults)
- M > F
- group 4 (most common)
- typically children (rare in infants)
- M:F 2:1
Clinical presentation
The growth of these very cellular tumors is often rapid and accounts for their relatively rapid clinical onset. Typically, presentation occurs over a few weeks with features that are dominated by symptoms of raised intracranial pressure as a result of obstructive hydrocephalus .
In approximately 40% of patients, there is evidence of CSF seeding at the time of diagnosis .
Pathology
The tumors, in general, tend to be extremely cellular and are an example of a small round blue cell tumor which results in predictable imaging features. They are categorized as CNS primitive neuroectodermal tumors (WHO grade IV).
Associations
Medulloblastomas are associated with a number of syndromes, including:
- Coffin-Siris syndrome
- Cowden syndrome
- Gardner syndrome
- Gorlin syndrome
- Li-Fraumeni syndrome
- Rubinstein-Taybi syndrome
- Turcot syndrome
- L-2-hydoxyglutaric aciduria
Radiographic features
The radiographic features are strongly influenced by the histological type and molecular subtype of the tumor. Many of the imaging characteristics can, however, be remembered by thinking of medulloblastoma as a small round blue cell tumor.
Overall the vast majority (94%) of medulloblastomas arise in the cerebellum and the majority of these, from the vermis (75%). They tend to protrude into the fourth ventricle from its roof, and may even grow directly into the brainstem . This pattern is particularly common in group 3 and group 4 and in some SHH subgroup tumors .
Other areas are less common and are seen more frequently in older children and adults. In such cases, the tumor is also more likely to be poorly marginated and demonstrate larger cyst formation . Adult medulloblastomas are usually located laterally, in the cerebellar hemispheres, with only 28% centered in the vermis; these are most commonly of the SHH subgroup .
Cerebellar peduncle epicenter is almost exclusively seen in the relatively indolent WNT subgroup .
CT
On CT, medulloblastomas often appear as a mass arising from the vermis, resulting in effacement of the fourth ventricle / basal cisterns and obstructive hydrocephalus. They can also occur more laterally in the cerebellum.
They are usually hyperdense (90%) and cysts formation/necrosis is common (40-50%), especially in older patients. Calcification is seen in 10-20% of cases .
Enhancement is present in over 90% of cases and is usually prominent .
MRI
- T1
- hypointense to grey matter
- T1 C+ (Gd)
- overall 90% enhance, often heterogeneously
- group 4 tumors tend to enhance less
- T2/FLAIR
- overall are iso to hyperintense to grey matter
- heterogeneous due to calcification, necrosis and cyst formation
- surrounding edema is common
- DWI/ADC
- high DWI signal ("restricted diffusion")
- low ADC values (lower than normal cerebellum e.g. ~550 x 10 mm/s)
- MR spectroscopy
- elevated choline
- decreased NAA
MRI is able to delineate the fourth ventricle and subarachnoid space to a much greater degree than CT. Although medulloblastomas project into the fourth ventricle, unlike ependymomas they do not usually extend into the basal cisterns .
As CSF seeding is common at presentation, imaging with contrast of the whole neuraxis is recommended to identify drop metastases and leptomeningeal spread. Although rare, extraneural spread is reported.
Predicting molecular subgroup from imaging
So if all this sounds confusing, that's because it is. Molecular subgroups, histology, location, appearance, and demographics all interact, but this notwithstanding, you can make some fairly robust predictions based on imaging when taking all of these together . Location is the key to this approach.
- cerebellar peduncle
- very likely WNT subgroup and therefore best prognosis
- cerebellar hemisphere
- very likely SHH subgroup and therefore intermediate prognosis
- likely desmoplastic/nodular/medulloblastoma with extensive nodularity (MBEN)
- midline
- may be group 3, group 4 or SHH
- typically infants with a tumor with ill-defined margins but prominent enhancement: likely group 3 (or SHH) and therefore worst prognosis
- typically children with a tumor with well-defined margins but mild or no enhancement: likely group 4 and therefore slightly better prognosis
- adults with variably defined and variably enhancing tumors: most likely SHH; hemorrhage raises the probability of group 4
MR spectroscopy may also be distinctive :
- group 3 or 4
- taurine peak
- high creatine
- SHH
- little or no taurine
- low creatine
Treatment and prognosis
Treatment typically consists of surgical resection, radiation therapy, and chemotherapy. In general, the tumors are quite radiosensitive.
Cerebellar mutism syndrome may occur after surgical resection of medulloblastoma .
Prognosis is most strongly influenced by molecular subtype :
- WNT: very good
- SHH: infants good, others intermediate
- group 3: poor
- group 4: intermediate
Traditionally, and still relevant in addition to molecular characteristics, prognosis if considered poor if young age at diagnosis (<3 years of age), incomplete surgical resection (>1.5 cm residual), and presence of CSF metastases at the time of diagnosis; common in infants and children (~25%) and uncommon in adults (~2%).
Histological features also have an effect on prognosis with desmoplastic and nodular histology having a better outlook than large cell or anaplastic histologic features .
Expression of the c-erbB-2 (HER2/neu) oncogene is useful in the staging of medulloblastomas. The increased c-erbB-2 expression reflects an increase in the proliferative activity of a tumor (widely used in breast cancer staging).
- no CSF metastases, complete surgical resection and negative c-erbB-2 expression: 5-year-survival 100%
- no CSF metastases, complete surgical resection and positive c-erbB-2 expression: 5-year-survival 54%
- CSF metastases and/or incomplete surgical resection: 5-year-survival 20%
History and etymology
In 1925, Bailey and Cushing described the first cases of medulloblastomas .
Differential diagnosis
In the pediatric population consider:
- ependymoma
- usually arises from the floor of the 4 ventricle
- typically squeezes out the foramen of Luschka
- does not usually cause as much diffusion restriction
- atypical teratoid/rhabdoid tumor
- very young children
- aggressive
- pilocytic astrocytoma
- usually cystic
- brainstem glioma (exophytic)
- choroid plexus papilloma (CPP): more common in lateral ventricles in children
In the adult population consider:

 Assoziationen und Differentialdiagnosen zu Medulloblastom:
Assoziationen und Differentialdiagnosen zu Medulloblastom:










