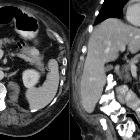
pancreatic ductal adenocarcinoma
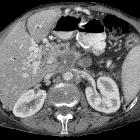


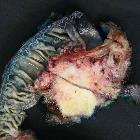
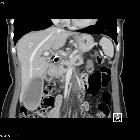
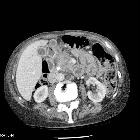
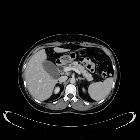
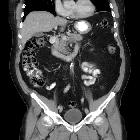

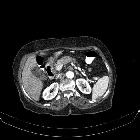
















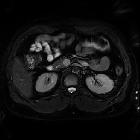

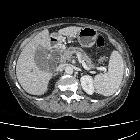





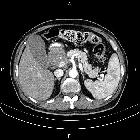




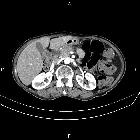
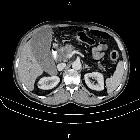
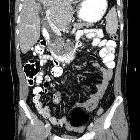
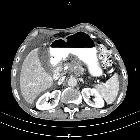

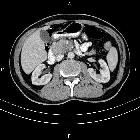







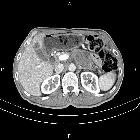


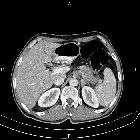

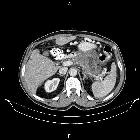





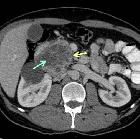
Pancreatic ductal adenocarcinoma makes up the vast majority (~90%) of all pancreatic neoplasms and remains a disease with a very poor prognosis and high morbidity.
On imaging, it usually presents as a hypodense mass on CT that is poorly marginated, which may encase vessels and the common bile duct.
Epidemiology
Pancreatic cancer accounts for 22% of all deaths due to gastrointestinal malignancy, and 5% of all cancer deaths . In general, it is a malignancy of the elderly with over 80% of cases occurring after the age of 60 .
Risk factors
Risk factors include:
- cigarette smoking: the strongest environmental risk factor
- a diet rich in animal fats and protein
- obesity
- family history: three or more first-order relatives with pancreatic cancer results in ~20x risk
- hereditary syndromes
Perhaps surprisingly there is only a weak if at all present association with heavy alcohol consumption .
Clinical presentation
- pain (most common)
- Courvoisier gallbladder: painless jaundice and enlarged gallbladder
- Trousseau syndrome: migratory thrombophlebitis
- new-onset diabetes mellitus
- lipase hypersecretion syndrome (10-15%)
- polyarthralgia and subcutaneous fat necrosis +/- lytic bone lesions
- elevated serum lipase and eosinophilia
Pathology
Three precursor lesions for pancreatic adenocarcinoma have been identified :
- pancreatic intraepithelial neoplasia (PanIN)
- intraductal papillary mucinous neoplasm (IPMN)
- mucinous cystic neoplasm
Cancerous cells arise from the pancreatic ductal epithelium. As the majority of tumors (90%) are not resectable, diagnosis is usually achieved with imaging (typically CT scan) although laparoscopy is often required to confirm resectability . The key to accurate staging is the assessment of the SMA and celiac axis, which if involved exclude the patient from any attempted resection .
Histological subtypes
- adenocarcinoma: majority
- acinar cell carcinoma of the pancreas
- adenosquamous carcinoma of the pancreas
- undifferentiated with osteoclasts giant cells
Location and classification
- head and uncinate process: two-thirds of cases
- body and tail: one-third of cases
Staging
Please see pancreatic ductal adenocarcinoma staging.
Recurrence is probably better estimated by a risk score than by staging
Radiographic features
Fluoroscopy
Barium meal/small bowel follow-through
If large enough may demonstrate a reverse impression on the duodenum: Frostburg inverted 3 sign or a wide duodenal sweep.
Ultrasound
Findings are non-specific and include:
- hypoechoic mass
- double duct sign may be seen
CT
CT is the workhorse of pancreatic imaging. Typically ductal adenocarcinomas appear as poorly defined masses with extensive surrounding desmoplastic reaction. They enhance poorly compared to adjacent normal pancreatic tissue and thus appear hypodense on arterial phase scans in 75-90% of cases, but may become isodense on delayed scans (thus the need for multiple phase scanning when pancreatic cancer is the clinical question). The double duct sign may be seen. Calcifications are very rare in adenocarcinoma and when present are more likely due to a pre-existing condition such as chronic pancreatitis .
CT correlates well with surgical findings in predicting unresectability (positive predictive value of 89-100% ). The most important feature to assess locally is the relationship of the tumor to surrounding vessels (SMA and celiac axis). If the tumor surrounds a vessel by more than 180 degrees, then it is deemed T4 disease and is unresectable .
MRI
Signal characteristics include:
- T1: hypointense cf. normal pancreas
- T1 FS: hypointense cf. normal pancreas
- T1 + C (Gd): slower enhancement than the normal pancreas, therefore dynamic injection with fat saturation with arterial phase imaging is ideal
- T2/FLAIR: variable (therefore not very useful), depending on the amount of reactive desmoplastic reaction
- MRCP: double duct sign may be seen
Treatment and prognosis
Most tumors are not resectable at diagnosis.
Surgery for stage I and II (see staging of pancreatic cancer) does offer the chance of cure, though with high morbidity (20-30%) and mortality (5%) . Resection is performed with a Whipple operation.
Even when resection is possible, the majority of patients succumb to recurrence, with only a doubling of survival in operated patients , from 5% to 10% at 5 years . At 12 months following the diagnosis, almost a quarter of the patients will have died .
Differential diagnosis
General imaging differential considerations include:
- acute pancreatitis
- chronic pancreatitis
- other pancreatic neoplasms
- lymphoma
- fatty infiltration of the pancreatic head
- usually involving the anterior portion
- no secondary signs (e.g. pancreatic duct or common bile duct dilatation)
- high signal on T1 and signal drop on chemical shift sequences
- cholangiocarcinoma
- periampullary tumors
- pancreatic metastases
Siehe auch:
- Insulinom
- Pankreastumoren
- zystische Pankreasläsionen
- intraduktale papillär muzinöse Neoplasie
- Zöliakusblockade
- Gastrinom
- solider pseudopapillärer Tumor des Pankreas
- neuroendokrine Pankreastumoren
- Pankreasmetastasen
- Pankreatoblastom
- Hamartom des Pankreas
- Lungenmetastasen Pankreaskarzinom
- Glukagonom
- Tuberkulose des Pankreas
- seröses Zystadenom
- mucinous cystic neoplasm
- zystischer neuroendokriner Tumor des Pankreas
- Riesenzelltumor des Pankreas
- mesenchymale Pankreastumoren
- papillary cystic neoplasm of the pancreas
- duktales Adenokarzinom
und weiter:
- kavernöse Transformation Pfortader
- Kerley-Linien
- Choledocholithiasis
- Pancreas anulare
- Akute Pankreatitis
- Autoimmunpankreatitis
- Krukenberg-Tumor
- cancer
- ultrasound appearances of liver metastases
- Sklerose der Papilla vateri
- Morbus Hippel-Lindau
- Maffucci-Syndrom
- intrapankreatische Nebenmilz
- solide Pankreastumoren
- antral pad sign
- whipple procedure
- bulging duodenal papilla
- Ikterus
- Karzinom der Papilla vateri
- TNM Staging Pankreaskarzinom
- Duodenalstent
- periampullary tumours
- Thrombose der Vena lienalis
- Kausch-Whipple-Operation
- Thrombose der Arteria splenica
- Mesenterialvenenenge
- ampullary tumour
- carcinoma of the head of the pancreas
- IPMN and pancreatic adenocarcinoma
- paraduodenale Pankreatitis
- inflammatorischer Pankreaspseudotumor
- Inselzellkarzinom
- Plattenepithelkarzinom des Pankreas

 Assoziationen und Differentialdiagnosen zu Pankreaskarzinom:
Assoziationen und Differentialdiagnosen zu Pankreaskarzinom: