Neurozystizerkose

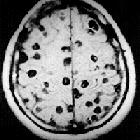


























Neurocysticercosis is caused by the CNS infection with the pork tapeworm Taenia solium, which is endemic in most low-income countries where pigs are raised. This form of cysticercosis is a relevant cause of seizures in endemic areas.
Epidemiology
The disease is endemic in Central and South America, Asia and Africa. The perpetuation of this parasitic disease is related to poor sanitation and hygiene.
There is no gender or race predilection and most symptomatic patients are aged 15-40 years .
Clinical presentation
There is a variable time interval between the point of infection and the onset of symptoms (ranging from 1-30 years).
Clinical presentation includes :
- seizures: most common symptom and the most common cause of seizures in young adults in endemic areas
- headaches
- hydrocephalus
- altered mental status
- neurological deficits
- Bruns syndrome: caused by cysticerci cysts of the third and fourth ventricle
CSF serology may be helpful with the initial diagnosis especially in cases of intraventricular/subarachnoid infection .
Pathology
Infection, which leads to extra-intestinal disease (including neurocysticercosis), usually occurs as a result of eating food or drinking water contaminated by human feces containing T. solium eggs. This is distinct from the 'normal' life cycle in which the undercooked pork is eaten and the larval cysts contained within, mature into adult intestinal tapeworm .
Extra-intestinal infection undergoes specific clinical and imaging changes at it progresses through four stages of infection .
Stages
There are four main stages (also known as Escobar's pathological stages):
Location
Infection can be both intra- and extra-axial. Commonest locations are :
- subarachnoid space over the cerebral hemispheres: can be very large
- parenchyma: most common location, frequently seen near the grey matter-white matter junction
- basal cisterns
- maybe "grape-like" (racemose): most lack an identifiable scolex
- ventricles
- usually solitary cyst
- 4 ventricle: most frequent location
- spinal forms: associated with concomitant intracranial involvement
Typically the parenchymal cysts are small (1 cm) whereas the subarachnoid cysts can be much bigger (up to 9 cm): differential, therefore, being an arachnoid cyst.
Radiographic features
Imaging findings depend on the location and stage of infection.
Location
Subarachnoid/intraventricular
When in the subarachnoid space/interventricular, the cysts typically do not have a visible scolex. In the basal cisterns, they can be grape-like (racemose). The cysts are typically 1-2 cm in diameter . Usually, the cysts are similar in signal intensity to CSF, although occasionally cyst fluid may somewhat differ .
In the ventricles, there is often (79%) associated ventriculitis often leading to aqueductal stenosis and hydrocephalus .
Parenchymal
Parenchymal cysts usually involve the grey-white matter junction .
Stage
Each stage has fairly distinctive imaging features although there is no sharp demarcation between later stages.
Vesicular
- cyst with dot sign
- CSF density/intensity
- eccentric hyperintense scolex on T1 can sometimes be seen
- no enhancement is typical, although very faint enhancement of the wall and enhancement of the scolex may be seen
- no surrounding vasogenic edema
Colloidal vesicular
- cyst fluid becomes macroscopically turbid reflected in altered imaging appearance:
- CT: hyperattenuating to CSF
- MRI T1: hyperintense to CSF
- surrounding edema
- cyst and the wall become thickened and brightly enhances
- scolex is seen early in the colloidal phase but gradually shrinks down and is harder to identify
Granular nodular
- edema decreases
- cyst retracts eventually becoming a small enhancing nodule
- enhancement persists but is less marked
Nodular calcified
- end-stage quiescent calcified nodule
- no edema
- no enhancement on CT
- signal drop out on T2 and T2* sequences
- some intrinsic high T1 signal may be present
- long term enhancement may be evident on MRI and may predict ongoing seizures
Treatment and prognosis
The treatment options available to patients with neurocysticercosis include symptomatic therapy (e.g. anti-epileptics) and anthelmintic therapy (e.g. albendazole and praziquantel, the two antiparasitics most commonly used ), usually accompanied by corticosteroids. Surgery (e.g. VP shunt placement or decompression) is only rarely indicated.
Differential diagnosis
General imaging differential considerations include:
- cerebral metastasis
- pyogenic cerebral abscess
- tuberculoma
- amoebic encephalitis
- perivascular spaces
- other parasitic/fungal infection(s)
Siehe auch:
- zerebrale Verkalkungen
- intrakranielle Verkalkungen
- Hirnmetastase
- Hirnabszess
- Hydrocephalus
- Virchow-Robin-Räume
- Aquäduktstenose
- Ventrikulitis
- Zystizerkose
- cyst with dot sign
und weiter:
- Arachnoidalzyste
- stark erweiterte Virchow-Robin-Räume
- Basalganglienverkalkungen
- lacunar infarct
- Sturge-Weber-Krabbe-Syndrom
- intraventrikuläre Neoplasien und Läsionen
- intrakranielle Epidermoidzyste
- Tuberkulose des ZNS
- Neurotoxoplasmose
- Brain stone
- neuroradiologisches Curriculum
- ependymal cyst
- intraventrikuläre Neoplasien und Läsionen - Überblick
- intraventricular epidermoid
- zerebrale Läsionen mit ringförmiger Kontrastmittelanreicherung
- intraaxial
- zerebrale Mikroangiopathie bei Hypertonus
- Tuberkulom des ZNS
- zerebelläre Zystizerkose
- Zystizerkose des Bulbus oculi
- supraselläre Arachnoidalzyste
- peri vascular space
- spinale Cysticercose
- NCC

 Assoziationen und Differentialdiagnosen zu Neurozystizerkose:
Assoziationen und Differentialdiagnosen zu Neurozystizerkose:







